Antibody screen negative8/7/2023  6 The patient's RBCs are placed at the top of the column in a reaction chamber and then centrifuged. 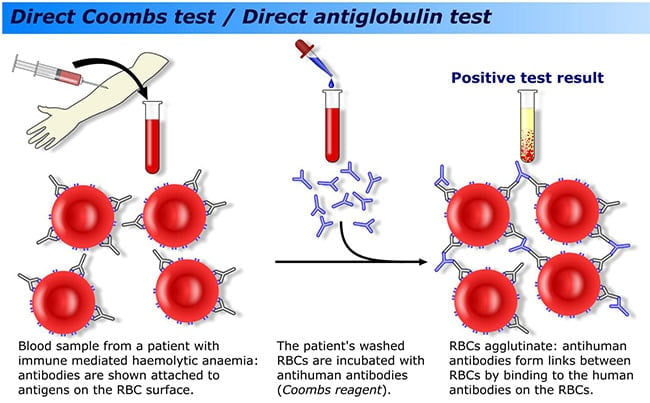
The gel method (examples include the ID-MicroTyping System by Ortho Diagnostics, Raritan, New Jersey and Diamed by BioRad, Hercules, California) uses a dextran acrylamide column that is impregnated with anti-human globulin reagent, trapping antibody-bound RBCs. In the United States, microcolumn technology is often referred to as the “gel” method. Microcolumn and solid phase are 2 commonly used alternatives to tube testing. Some advancements above and beyond this tube method have been introduced in immunohematologic testing, and these platforms have been applied to the DAT. The test tubes can then be examined visually for agglutination and graded from 0 to 4+, with 4+ representing a solid button of agglutination. If a complement reagent is used, and if the reagent manufacturer's package insert permits, there may be a second incubation to allow enhancement of weak anti-complement reactions. 5 After addition of the polyspecific reagent, the RBCs are centrifuged. Both inadequate RBC washing and/or a delay in test performance can lead to a false-negative result. After washing, it is important to proceed as soon as possible to testing before any bound antibody has a chance to disassociate (or elute) off the patient's RBCs. 5 In the classic tube testing version of the DAT, the RBCs to be tested should be washed with saline to remove excess, unbound IgG or complement present in the serum that could preferentially bind and inhibit the reactivity of the added reagent. EDTA is necessary to chelate calcium (a necessary component of C3 activation) so that in vitro C3 fixation will not occur. The appropriate specimen for the DAT is one that is anticoagulated with ethylenediaminetetraacetic acid (EDTA). BASICS OF DAT PERFORMANCE AND AVAILABLE TESTING PLATFORMS 3 Since this discovery, the DAT has become a critical tool in the assessment of both immune and drug-induced hemolytic anemias, HDFN, as well as a fundamental step in the evaluation of patients experiencing transfusion reactions. It was 1 year later that he published the use of the DAT as a method of demonstrating in vivo sensitization of RBCs in hemolytic disease of the fetus and newborn (HDFN). 
4 That antibody became the now commonly used anti-human globulin that uses the 2 Fab sites of the reagent antibody to bind the Fc portion of the antibody that is coating target RBCs. He suddenly recognized he could use a second antibody formed to the RBC coating antibody that would “bridge” and cause visible RBC agglutination. 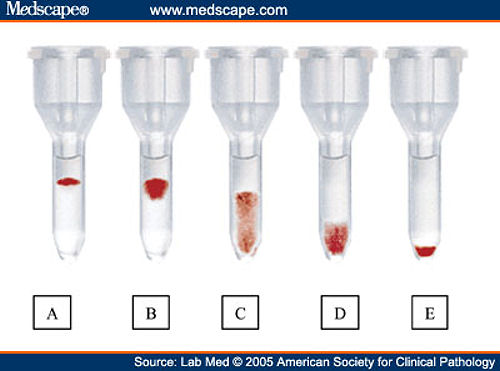
3 Questioning how to measure these “incomplete” antibodies, Coombs realized the answer during a long delayed wartime train trip from London to Cambridge. Furthermore, the RBCs were no longer able to bind with their respective “complete” or agglutinating antibody. 2 When testing patient plasma for the presence of Rh antibody with the use of Rh antigen–positive RBCs, it was observed that sometimes the RBCs appeared to be sensitized by the respective Rh antibody, but did not result in agglutination. The principle of using anti-human globulins was first described by Moreschi in 1908, 1 but it was not until 1945 that Robin Coombs introduced it to clinical medicine initially as a method to demonstrate RBC agglutination in the presence of what was then thought to be an “incomplete” or “blocking” antibody as seen within the context of the IAT. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
